NEW YORK (Reuters Health)—Subcutaneous belimumab improves outcomes in anti-dsDNA-positive hypocomplementemic patients with systemic lupus erythematosus (SLE), according to results from a Phase 3 randomized, controlled trial. Intravenous belimumab is approved by the U.S. Food and Drug Administration and the European Medicines Agency for the treatment of patients with active, autoantibody-positive SLE who are receiving standard…
Search results for: belimumab

European Commission Approves Subcutaneous Belimumab for SLE; FDA Safety Alert for Febuxostat
Subcutaneous belimumab has been approved in the EU to help treat patients with active autoantibody-positive SLE…

OA Knee Pain Treatment Enters Clinical Trials, Ixekizumab Receives FDA Approval & Belimumab Promising for SLE Patients
Clinical trials have begun to determine if disodium zoledronate tetrahydrate is safe and effective for treating pain in patients with knee osteoarthritis. The FDA has approved the use of ixekizumab for the treatment of plaque psoriasis. And a study has shown that patients with SLE treated with belimumab may be able to decrease steroid use…
Drug Updates: Apremilast, Belimumab, and More
Information on new approvals and medication safety
August Updates from the ACR Insurance Subcommittee
The Insurance Subcommittee of the Committee on Rheumatologic Care is working on behalf of ACR members to address critical coverage and reimbursement challenges.

Emerging Therapies for Antiphospholipid Syndrome, an Antibody-Mediated Disease
A recent A&R review explores innovative, emerging therapies for the currently incurable, antibody-mediated disease antiphospholipid syndrome.

Rheuminations: Can We Now Control the Wolf?
Lupus has always had a reputation for being a wild, unrestrained and enigmatic entity. In fact, the very name lupus comes from the Latin word for wolf, a gift from our Roman predecessors who saw a resemblance between lupus rashes and a wolf’s bite. Given the limitations of immunology back then, it is an incredibly…
FDA Approvals: Lupus Nephritis
Lupus nephritis is one of the leading causes of mortality for patients with systemic lupus erythematosus (SLE), and patients with both SLE and end-stage renal disease have standardized mortality ratios more than 60 times that of patients with SLE with normal kidney function.1 The good news: Rheumatologists now have not one, but two approved options…
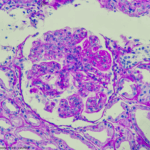
The ACR Releases New Lupus Nephritis Guideline
At a session of ACR Convergence 2024, speakers shared key elements of the new guideline on the screening, treatment and overall management of lupus nephritis in children and adults.1 This guideline attempts to balance the risks of medication side effects with the important goal of preserving kidney function. This is the ACR’s first lupus guideline…

B Cell-Depleting Therapy in SLE & Lupus Nephritis
“B cells are central orchestraters of autoimmune disease by contributing to autoantibody production, antigen presentation and cytokine production,” says Georg Schett, MD, head of the Department of Medicine 3 (Rheumatology and Immunology) of the Friedrich-Alexander University Erlangen-Nürnberg, Germany. “B cells can be inhibited by monoclonal antibodies and chimeric antigen receptor (CAR) expressing cells.” Dr. Schett…
- « Previous Page
- 1
- 2
- 3
- 4
- …
- 15
- Next Page »