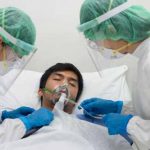
The FDA has authorized a phase 3 clinical trial examining the safety and efficacy of tocilizumab plus standard care in patients hospitalized with COVID-19…


The FDA has authorized a phase 3 clinical trial examining the safety and efficacy of tocilizumab plus standard care in patients hospitalized with COVID-19…
Afton Hassett, PsyD, offers self-care tips and resources to handle stress, avoid burnout and stay healthy while caring for others.
Reuters Staff |
NEW YORK (Reuters Health)—The ongoing novel coronavirus pandemic has raised concerns about whether biologic therapy could make psoriasis patients more susceptible to SARS-CoV-2, the virus behind COVID-19. Mark Lebwohl, MD, from Icahn School of Medicine at Mount Sinai Hospital in New York and colleagues address this issue in a letter in the Journal of the…
Matthias Blamont & Michael Erman |
PARIS (Reuters)—Sanofi SA will be able to provide millions of doses of hydroxychloroquine (HCQ) for patients with the illness caused by the novel coronavirus if the old malaria drug proves successful in clinical trials, its chief executive tells Reuters on Thursday. Paul Hudson, who became CEO of the French drugmaker in September, said in an…
The Centers for Medicaid & Medicare Services (CMS) announced last week an extension of the 2019 Merit-Based Incentive Payment System (MIPS) reporting deadline to 8 p.m. EDT on Thursday, April 30. The extreme and uncontrollable circumstances policy has also been updated to automatically apply to any MIPS-eligible providers who do not submit data by the…
The outbreak of the SARS-CoV-2 virus is disrupting rheumatology clinics and practices all over the world. In the U.S., many providers are turning to telemedicine to care for their patients, but struggles remain. Philip A. Waller, MD, who practices at Accurate Clinical Research, Houston, spoke with The Rheumatologist about how the pandemic is affecting his…
Rheumatology practices are being hit hard by the COVID-19 pandemic. The sustainability of practices is being threatened and patient access to care is being reduced. The $2 trillion Coronavirus Aid, Relief and Economic Security (CARES) Act has numerous provisions that bring much-needed support to physicians and their practices. This includes $100 billion for healthcare providers, including physician…

The new landscape for rheumatologists includes telemedicine, kids out of school and infection-prevention protocols. During the current pandemic, the practice of medicine, research and daily lives are changing to keep providers and patients safe…
Reuters Staff |
(Reuters)—Sanofi and Regeneron Pharmaceuticals Inc. have expanded a clinical trial of their rheumatoid arthritis drug Kevzara (sarilumab) as a coronavirus treatment to patients outside the U.S. The companies said enrolments for the mid-to-late stage trial of sarilumab, an immune-system modifying drug known as a monoclonal antibody, have now started in Italy, Spain, Germany, France, Canada…
Amish J. Dave, MD, MPH |
A timely and personal appeal to all those who can to become or remain civically active in this critical time.