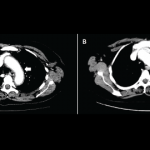
A 53-year-old female presented to the clinic for severe polyarticular joint pain and was found to have a seronegative inflammatory arthritis. Six months before, she had completed 10 months of treatment for stage IV metastatic melanoma with the immune checkpoint inhibitors, nivolumab and ipilimumab, achieving complete remission of her cancer.
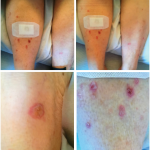
She said that throughout her course of immunotherapy treatment she had diffuse aching in her large joints, especially on the days immediately after her infusions, as well as dry eyes and dry mouth. Her course was complicated by several adverse events, including rash and colitis, for which she received prednisone ranging from 20–60 mg daily. By the time she had completed her last infusion of nivolumab, the joint pain and stiffness had spread to the small joints of her hands.
After our evaluation showed synovitis in the small joints of the hands, wrists and knees, she was started on methotrexate and prednisone, with improvement in her arthritis disease activity over several months. Her sicca syndrome remains active, requiring frequent use of replacement eye drops. Her melanoma remains in remission.
Immune Checkpoint Inhibitors
Immune checkpoint inhibitors (ICIs) have dramatically changed the face of cancer treatment, and in 2013, Science magazine deemed cancer immunotherapy the “breakthrough of the year.”1 ICIs have shown clinical benefit for the treatment of advanced cancers, including melanoma, non-small/small cell lung cancer, renal cell carcinoma, Hodgkin’s lymphoma, head and neck cancers, and urothelial cell carcinoma.
Numerous clinical trials are underway to evaluate ICI efficacy in a variety of other tumor types and in earlier stages of disease. The side effects of ICIs are related to their mechanism of action. ICIs cause nonspecific immune activation, which can target non-tumor tissues, thus leading to immune-related adverse events (irAEs). irAEs can resemble various rheumatic diseases, such as inflammatory arthritis and sicca syndrome, but also include diverse manifestations, such as rash, gastrointestinal (GI) disturbance and endocrinopathies. As indications for immune checkpoint blockade expand and ICIs are used in combination, it becomes increasingly important for rheumatologists and rheumatology professionals to recognize irAEs, their connection to cancer immunotherapy and how to treat these events appropriately.
Timeline of Immunotherapies, Mechanisms of Action
Cancer immunotherapy has shifted the current management strategy in advanced cancer from conventional chemotherapy approaches to using endogenous immune cells to target cancer. ICIs are a novel class of immunotherapies, first approved by the FDA in 2011. Several drugs are currently on the market, including ipilimumab, nivolumab, pembrolizumab and atezolizumab; many more ICIs are in clinical trials (see Table 1).