SAN DIEGO—In a session titled, In Vivo Administration of MiR-146a Protects C57BL/6 Mice from Pristane-Induced Pulmonary Hemorrhage via Suppressing Type 1 Interferon Response, Nan Shen, MD, professor of medicine and director, Shanghai Institute of Rheumatology, Renji Hospital, Jiao Tong University School of Medicine, Shanghai, China, presented the Rheumatology Research Foundation Edmond L. Dubois, MD, Memorial Lectureship at the 2013 ACR/ARHP Annual Meeting. (Editor’s note: This session was recorded and is available via ACR SessionSelect at www.rheumatology.org.)
Along with reviewing previous studies that have shown the crucial role of microRNAs (miRNAs) for human lupus, and in particular the role of a novel functional variant in the microRNA-146a gene, Dr. Shen presented findings from a current study that explored a therapeutic potential of miRNA-146a in a murine model of lupus.
“Growing evidence shows deregulation and dysfunction of miRNAs in human lupus and lupus-like mice model, and that miRNAs are associated with prognosis and progression of several human diseases and may serve as future targets for gene therapy,” Dr. Shen said. “Indeed, the clear importance of miRNA to disease expression is striking in both man and mouse, and several clinical trials are now underway using [a] miRNA inhibition approach for treating several common diseases.”
MicroRNAs—Some Background
“MicroRNAs represent a large class of small non-coding RNAs that ubiquitously exist in a broad range of species from worms to mammals, which function as an important class of molecular mediators functioning in post-transcriptional regulation, [and] play a crucial role in diverse biological processes, including regulation of immune response and involvement in autoimmunity,” said Dr. Shen.
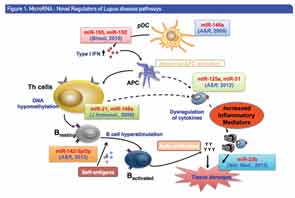
Figure 1 illustrates miRNAs as novel regulators of the disease pathways of lupus. In his talk, Dr. Shen provided data from previous studies he and his colleagues have done to dissect the molecular mechanisms underlining the genetic and epigenetic control of lupus development. In these studies, they were able to show that overexpression of miRNA-146a in the peripheral blood mononucleated cells (PBMCs) from patients with systemic lupus erythematosus (SLE) suppressed activation of the type 1 interferon (IFN) pathway.1,2,3 This finding suggested to Dr. Shen and his colleagues that manipulation of miRNA-146a levels may be a useful therapeutic intervention in SLE, and prompted the current study to investigate the therapeutic potential of miRNA-146a for lupus intervention by an induced murine model of SLE.
Study: Therapeutic Potential of miR-146a in Induced Murine Model of Lupus
In their current study, Shen and colleagues used pristane-induced lupus, a well-established SLE murine model featuring abnormal activation of IFN pathway and an unusually high incidence of pulmonary capillaritis. Mice were intravenously injected with MiRNA-146a or phosphate-buffered saline (PBS) via lateral tail veins. Real-time polymerase chain reaction (PCR) or gene expression profile analysis was used to evaluate the peripheral blood drawn 3, 7 and 14 days after pristane injection. Mice were categorized into three groups for comparison: those receiving three consecutive injections of PBS (control group; n=9), those receiving miRNA-146a three days before a single intraperitoneal injection of pristane (prevention group; n=19), and those receiving miRNA-146a seven days after a single intraperitoneal injection of pristane (treatment group; n=10). The development of pulmonary hemorrhage was confirmed two weeks after the pristane injection using a hematoxylin and eosin (HE) stain.