In late October, the FDA approved the first biosimilar to ustekinumab to treat patients with psoriasis, psoriatic arthritis and other conditions. Ustekinumab-auub is expected to be available in the U.S. by 2025.


In late October, the FDA approved the first biosimilar to ustekinumab to treat patients with psoriasis, psoriatic arthritis and other conditions. Ustekinumab-auub is expected to be available in the U.S. by 2025.
Adil Vural, MD, & Kinanah Yaseen, MD |
Granulomatosis with polyangiitis (GPA) is a primary, necrotizing granulomatous vasculitis, involving small- to medium-sized arteries, that causes systemic disease. Almost any organ can be affected, but the most affected systems are the upper airways, lungs, kidneys, eyes and peripheral nerves. Migratory polyarthritis is reported in approximately 25% of patients with anti-neutrophil cytoplasmic antibody (ANCA) associated…
Saud Abaalkhail, MD, Muhammad Umair Javaid, DO, & Amarie Negron Rodriguez, MD |
Systemic autoinflammatory diseases (SAIDs) are rare syndromes characterized by alterations in innate immunity that result in a variety of clinical manifestations that are usually associated with recurrent fevers.1 Thanks to advances in genetic sequencing over the past few years, monogenic causes for some of these autoinflammatory diseases, such as Yao syndrome, have been discovered.2 Previously…
PHILADELPHIA—Officials from the U.S. Food & Drug Administration discussed recent drug approvals and drug safety issues at ACR Convergence in November in a session that captured the flurry of activity in the rheumatology sphere at the agency over the past year. Sabiha Khan, MD, clinical reviewer in the Division of Rheumatology and Transplant Medicine at…

After examining study data from pediatric patients with both psoriasis and psoriatic arthritis (PsA), the FDA approved ustekinumab as a treatment for patients aged 6–17 years old with PsA.
A prospective observational study by Syversen et al. found that patients with immune-mediated inflammatory diseases (IMID) had an attenuated serologic response to the standard two-dose vaccine regimen but a third dose was safe and effective.

Katie Robinson |
Findings support the continued use of TNF inhibitor monotherapy in individuals with immune-mediated inflammatory diseases. In the study, these patients had a lower risk of hospitalization or death caused by COVID-19 than patients on other commonly prescribed treatment regimens
Maria de la Luz Garcia-Hernandez, PhD, & Christopher Ritchlin, MD, MPH |
The etiology of psoriatic arthritis (PsA) is poorly understood but current evidence supports an interaction between genetic and environmental factors that coalesce to promote local tissue inflammation.1-3 The pivotal cytokines that underlie the local inflammatory response in a wide range of tissues are interleukin (IL) 23, IL-17 and tumor necrosis factor (TNF).4 The central contribution…
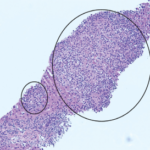
Osman Bhatty, MD, Dale Kobrin, MD, Lauren Mathos, DO, Nazia Khatoon, MD, Yazan Samhouri, MD, Naga Sai Krishna Patibandla, MD, & Mary Chester Wasko, MD, MSc |
A 26-year-old woman presented to our emergency department (ED) with intermittent fevers, nausea and vomiting. She had a past medical history of well-controlled, anti-nuclear antibody positive and rheumatoid factor negative polyarticular juvenile idiopathic arthritis (pJIA) and Crohn’s disease. Her maintenance treatment consisted of monthly intravenous infliximab, 10 mg of oral methotrexate weekly and 20 mg…
In February A&R, Penso et al. reported on the results of their study, which explored whether patients with psoriasis, PsA and AS have a higher risk of developing IBD when treated with an IL-17 inhibitor compared with apremilast, a phosphodiesterase 4 (PDE4) inhibitor, or etanercept, a TNF inhibitor.