NEW YORK (Reuters Health)—Gastrointestinal side effects are the most common reason osteoporotic women cite for nonadherence to oral bisphosphonate therapy, according to a new survey. “Our findings highlight the importance of low tolerability to nonadherence with osteoporosis therapy and underlines patients’ poor awareness and suboptimal physicians’ involvement in conveying the importance of this therapy,” Dr….
Search results for: alendronate
Rheumatology Coding Corner Question: Office Visit with DEXA Scan
A 67-year-old female patient with Medicare returns to the office for a follow-up of her age-related osteoporosis. She states she has an achy pain in her left hip that lasts for 30–40 minutes in the morning. Currently, she has taken ibandronate sodium and alendronate sodium for the past year, and her pain level is a…
New Bisphosphonate Therapy Recommendations for Postmenopausal Osteoporosis
A task force of the American Society for Bone and Mineral Research (ASBMR) has released new recommendations delineating the potential benefits and risks of prolonged therapy with oral and IV bisphosphonate therapy and providing guidance on duration of bisphosphonate therapy for postmenopausal osteoporosis.1 The task force makes clear that data and clinical experience on which…
2015 ACR/ARHP Annual Meeting: Research Lends Insight Into Osteoporosis Treatment, New Auto-Inflammatory Disease, Scleroderma
SAN FRANCISCO—Post-menopausal women with osteoporosis, previously treated with oral bisphosphonates, had greater increases in bone density when taking denosumab compared with zoledronic acid over a year’s time, according to a study presented at the 2015 ACR/ARHP Annual Meeting. The findings were discussed in the Discovery 2015 plenary session, which focused on new research. In the…
Denosumab Does Not Stimulate Early Bone Formation
NEW YORK (Reuters Health)—The early elevation in intact parathyroid hormone (iPTH) seen with denosumab treatment is not associated with increased bone formation, according to research from Eli Lilly and Company. Denosumab is a member of the anticatabolic/antiresorptive class of drugs used to treat osteoporosis; iPTH levels are increased in the first several months after denosumab…

Looking Back on Rheumatology in 2015, Leaping Forward to the Year Ahead
My dear friends, we come to praise Caesar. As we march toward 2016, we ought to acknowledge the great Roman emperor’s role in creating a proper calendar. At the start of Caesar’s reign, the calendar year lasted 355 days, 10¼ days fewer than the time it took the earth to fully orbit the sun.1 Although…

FDA Issues Stronger Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) Warning
The U.S. Food and Drug Administration (FDA) has toughened the existing warnings for nonsteroidal anti-inflammatory drugs (NSAIDs) due to their stroke and myocardial infarction (MI) risk increase.1 Due to a continual review of these products, FDA is requiring label updates for all prescription NSAIDs. Over-the-counter (OTC) NSAIDs already list the increased risk of MI and…
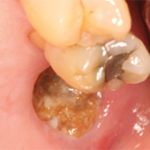
Diagnosis, Management of Medication-Induced Osteonecrosis of the Bone
Prior to the widespread use of bisphosphonates for the management of osteoporosis, multiple myeloma and metastatic cancer to the skeletal system, osteonecrosis of the jawbones was an infrequent condition seen after radiation for oral cancers (osteoradionecrosis) and in chronic odontogenic infections.1 Since the mid-2000s, osteonecrosis of the jawbones has been noted to occur as a…
Abaloparatide-SC May Reduce Fractures for Osteoporosis & New FDA Safety Website
In a 25 month Phase 3 trial, abaloparatide-SC reduced the risk of new fractures in patients suffering from postmenopausal osteoporosis. Plus, the FDA launches a new drug safety website.
Did Reports of Side Effects Contribute to Drop in Bone Drug Use?
(Reuters Health)—Media reports raising safety concerns about osteoporosis drugs known as bisphosphonates may have contributed to a sharp drop in their use—even though U.S. doctors and drug regulators haven’t recommended against taking them, a study suggests. Fosamax (alendronate sodium) won U.S. marketing approval in 1995. Widespread use of the drug and others like it over…
- « Previous Page
- 1
- …
- 3
- 4
- 5
- 6
- 7
- 8
- Next Page »