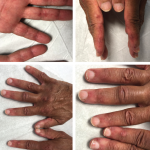
ACR CONVERGENCE 2020—Held Nov. 5–9, the ACR’s first fully virtual annual meeting provided participants with a vast repository of new research related to rheumatoid arthritis (RA). To help you sort through the noise, Elizabeth (Blair) Solow, MD, an assistant professor of medicine in the Division of Rheumatic Diseases at UT Southwestern Medical Center, Dallas, offered …