Thomas Dörner, MD, reviewed the current state of systemic lupus erythematosus (SLE) management, providing updates on novel therapies and insights into the pathogenesis of SLE.

Thomas Dörner, MD, reviewed the current state of systemic lupus erythematosus (SLE) management, providing updates on novel therapies and insights into the pathogenesis of SLE.

Clinicians have numerous treatment options for SLE; in a EULAR 2022 session, some of the newer therapies were reviewed.
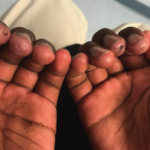
Anusha Vuppala, MD, & Sarwat Umer, MD |
Juvenile dermatomyositis (JDM) is a childhood form of inflammatory myopathy affecting children younger than 18 years. The diagnosis of JDM is based on characteristic clinical features of proximal muscle weakness, skin rash, elevated muscle enzymes, magnetic resonance imaging and muscle biopsy.1 Autoantibodies seen in patients with inflammatory myopathy are not only associated with particular phenotypes…

Upadacitinib is an investigational, oral, small molecule JAK1-selective inhibitor, which recently completed phase 3 clinical trials for treating patients with moderate to severe rheumatoid arthritis (RA). In late 2018, AbbVie Inc. submitted a New Drug Application to the U.S. Food and Drug Administration, as well as a marketing authorization application to the European Medicines Agency,…

CHICAGO—Although rare, when a patient has both primary immune deficiency and autoimmune disease, the combination can lead to life-threatening complications requiring careful, long-term therapy. In When Immune Deficiency and Autoimmunity Coexist, a session at the 2018 ACR/ARHP Annual Meeting, M. Eric Gershwin, MD, the Jack and Donald Chia professor of Medicine and chief of Rheumatology,…

When a patient has both primary immune deficiency and autoimmune disease, the combination can lead to life-threatening complications. Here are some insights into the challenges of diagnosing and treating this rare subset of patients…

The FDA has approved halobetasol propionate lotion as a topical treatment for plaque psoriasis…

Initial results from an ongoing trial show that upadacitinib outperforms adalimumab in achieving ACR20 in patients with rheumatoid arthritis…

FDA Approves Adalimumab-adbm On Aug. 29, the FDA approved Cyltezo (adalimumab-adbm), a biosimilar to Humira (adalimumab).1 Cyltezo was approved as a prefilled syringe to treat multiple chronic inflammatory diseases, including moderate to severe active RA, active psoriatic arthritis and ankylosing spondylitis, and moderate to severe plaque psoriasis. The treatment has also been approved for moderate…
Take the Challenge. ICD-10 Codes R76.1—Raised antibody titer L20.8—Other atopic dermatitis R20.2—Paresthesia of skin R20.1—Hypoesthesia of skin CPT Codes 99358 and 99359 Rationale The Centers for Medicare & Medicaid Services (CMS) typically does not allow separate payment for physician services that do not require face-to-face time with a patient, but as of Jan. 1, 2017,…