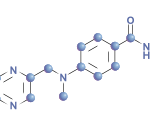
Research has found that taking adalimumab plus methotrexate is effective for patients with juvenile idiopathic arthritis-associated uveitis. However, these patients may also experience an increased incidence of adverse and serious adverse events compared with those taking only methotrexate…