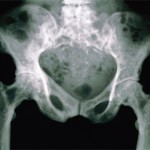
CHICAGO—A 49-year-old woman has had RA for eight years. She has a rheumatoid factor reading of 35, an aCCP reading of 160, erythrocyte sedimentation rate of 42, plus erosions. She has been on methotrexate. She tried etanercept for six months, but then it stopped working. She was on 40 mg of adalimumab weekly, but it…