According to a German study presented during ACR Convergence 2021, treatment persistence with JAK inhibitors is comparable to TNF inhibitors and other bDMARDs among patients with rheumatoid arthritis.


According to a German study presented during ACR Convergence 2021, treatment persistence with JAK inhibitors is comparable to TNF inhibitors and other bDMARDs among patients with rheumatoid arthritis.

Jeffrey Curtis, MD, MS, MPH |
ACR CONVERGENCE 2021—The ACR Convergence 2021 meeting reflected the continued advancement of science and practical research in the field of rheumatoid arthritis (RA). Among the most important topics this year in RA was the evolution of the risk-benefit profile of Janus kinase (JAK) inhibitors, for which new safety data emerged in a series of related…
Options to escalate treatment for refractory inflammatory myositis and new therapies in the pipeline are discussed.

Elizabeth Sloan, MD |
ACR Convergence 2021—The Pediatric Rheumatology Year in Review began with a fascinating talk by Mara Becker, MD, MSCE, a professor of Pediatrics and vice chair of faculty at Duke University School of Medicine, Durham, N.C. Dr. Becker began by describing her search strategy to select high-yield clinical science publications in pediatric rheumatology from the past…

ACR Convergence 2021—The recent Boxed Warning requirement applied to three Janus kinase (JAK) inhibitors, cautioning doctors and patients about several major risks in patients with rheumatoid arthritis, came only after rigorous data collection and careful consideration of the risks and the benefits, U.S. Food & Drug Administration (FDA) officials said in November at ACR Convergence…
ACR Convergence 2021—On Nov. 5, Karen H. Costenbader, MD, MPH, professor of medicine, Harvard Medical School, and director of the lupus program, Brigham and Women’s Hospital, Boston, gave a whirlwind review of the most important clinical rheumatology publications of the past year. Testing New Medications for Rheumatic Disease ADVOCATE Trial of Avacopan Dr. Costenbader first…

A key question many graduating rheumatology fellows face each year is: Are you interested in pursuing a career in academic medicine or in private practice? Although the two tracks are not mutually exclusive, it is true that juggling the demands of scholarly work, medical education and a busy clinical workload is by no means easy….
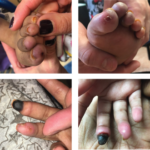
Marta Michalska-Smith, MD, & Colleen K. Correll, MD, MPH |
Janus kinase 1 and 2 inhibitors (jakinibs) have been approved for the treatment of rheumatoid arthritis, psoriatic arthritis and, most recently, juvenile idiopathic arthritis. They have also shown promise in the treatment of interferon (IFN) mediated diseases. The Janus kinase and signal transducer and activator of transcription (JAK/STAT) pathway is the principal signaling pathway for…

On Sept. 1, the U.S. Food & Drug Administration (FDA) announced that it is requiring revisions to the Boxed Warning for the Janus kinase (JAK) inhibitors Xeljanz/Xeljanz XR (tofacitinib), Olumiant (baricitinib) and Rinvoq (upadacitinib) to include information about the risks of serious heart-related events, cancer, blood clots and death.1 Recommendations for healthcare professionals will include…

In a study, Janus kinase inhibitors proved effective for patients with difficult-to-treat rheumatoid arthritis (RA).