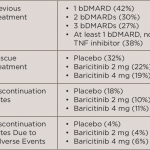
Soon, rheumatologists may have another drug to offer their patients with refractory rheumatoid arthritis (RRA) for whom effective and safe treatment remains challenging. A study published in the New England Journal of Medicine shows that patients with RRA treated with once-daily baricitinib in a 4 mg dose had a significant clinical improvement in symptoms of…